plant lab
My lab partner Kate and I did a lab where we were seeing if the plants we used were a good medicine, or not.
Objective/Purspose:
To find out which plants in our area contain active ingridents that will inhibit bacteria growth.
Materials:
balance, weight boat, lab scoops
LB broth base
media bottles, 250 ml
sterilizer/autoclave
water bath, 37* C, shaking sterile LB agar
laminar flow hood and disinfectant
glasses, safety, plastic
bunsen burner
gas lighter
inoculating loop, Ni/Cr wire
petri dishes, 60x15 mm, sterile E. coli JM109 (stock pile)
plant specimen
motar and pestle
pipet, 10 ml and pump
plastic funnels, short-stemmed
filter paper disks, 5 mm diameter
beakers, 100 ml
Objective/Purspose:
To find out which plants in our area contain active ingridents that will inhibit bacteria growth.
Materials:
balance, weight boat, lab scoops
LB broth base
media bottles, 250 ml
sterilizer/autoclave
water bath, 37* C, shaking sterile LB agar
laminar flow hood and disinfectant
glasses, safety, plastic
bunsen burner
gas lighter
inoculating loop, Ni/Cr wire
petri dishes, 60x15 mm, sterile E. coli JM109 (stock pile)
plant specimen
motar and pestle
pipet, 10 ml and pump
plastic funnels, short-stemmed
filter paper disks, 5 mm diameter
beakers, 100 ml
syringe, 10 ml and filter, 0.2 um
reaction tubes and rack, 1.7 ml
methanol, absolute pipet
pipet, 1 ml and pump
dry block heater/heat block
forceps, fine-tipped
ampicillin
glass spreader
incubator oven, 37 degrees celsius
reaction tubes and rack, 1.7 ml
methanol, absolute pipet
pipet, 1 ml and pump
dry block heater/heat block
forceps, fine-tipped
ampicillin
glass spreader
incubator oven, 37 degrees celsius
Procedure:
Prepare the plant extracts:
Prepare the plant extracts:
- using a mortar and pestle, grind up two grams of plant tissue with 10mL of deionized water
- let it set for three minutes
- filter the sample through am 11 cm filter paper/funnel
- filter sterilize the extract using a syringe filter
- collect 1 mL of the filter-sterilized extract into a 1.7 microbe
- label the sample
- attach pre filters to syringe and rinse with water
- take plant extracts, syringe, prefilter, and pipet to laminar hood
- label micro fuge tube W(water) & M(methanol)
- attach sterile filter to pre filter
- load 1.7 mils of extract into syringe, using pipet
- depress the plunger
- use less thank 1.0 ml filter-sterilization extract
- snap cap on micro fuge tube
preparing agar plates:
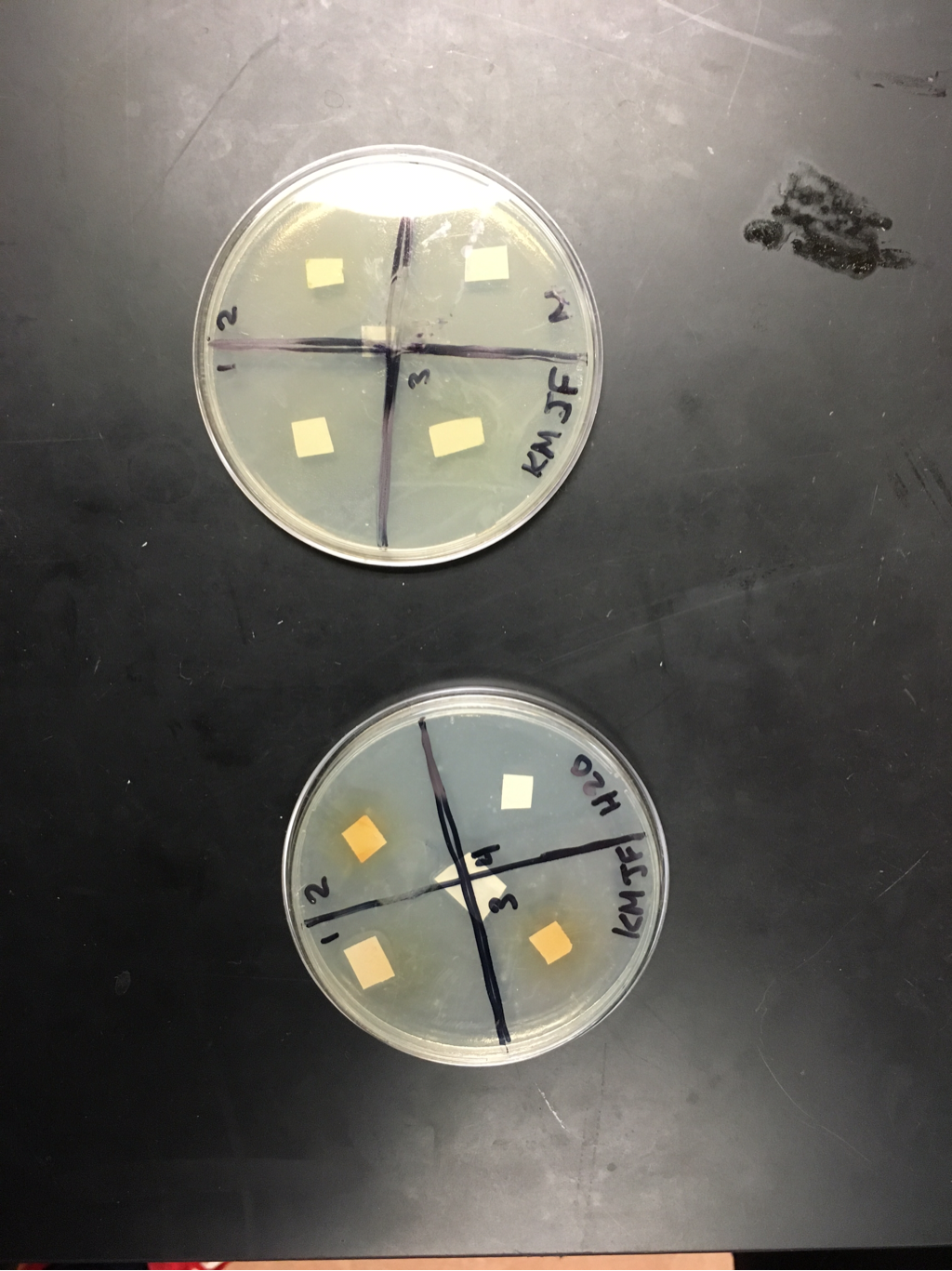
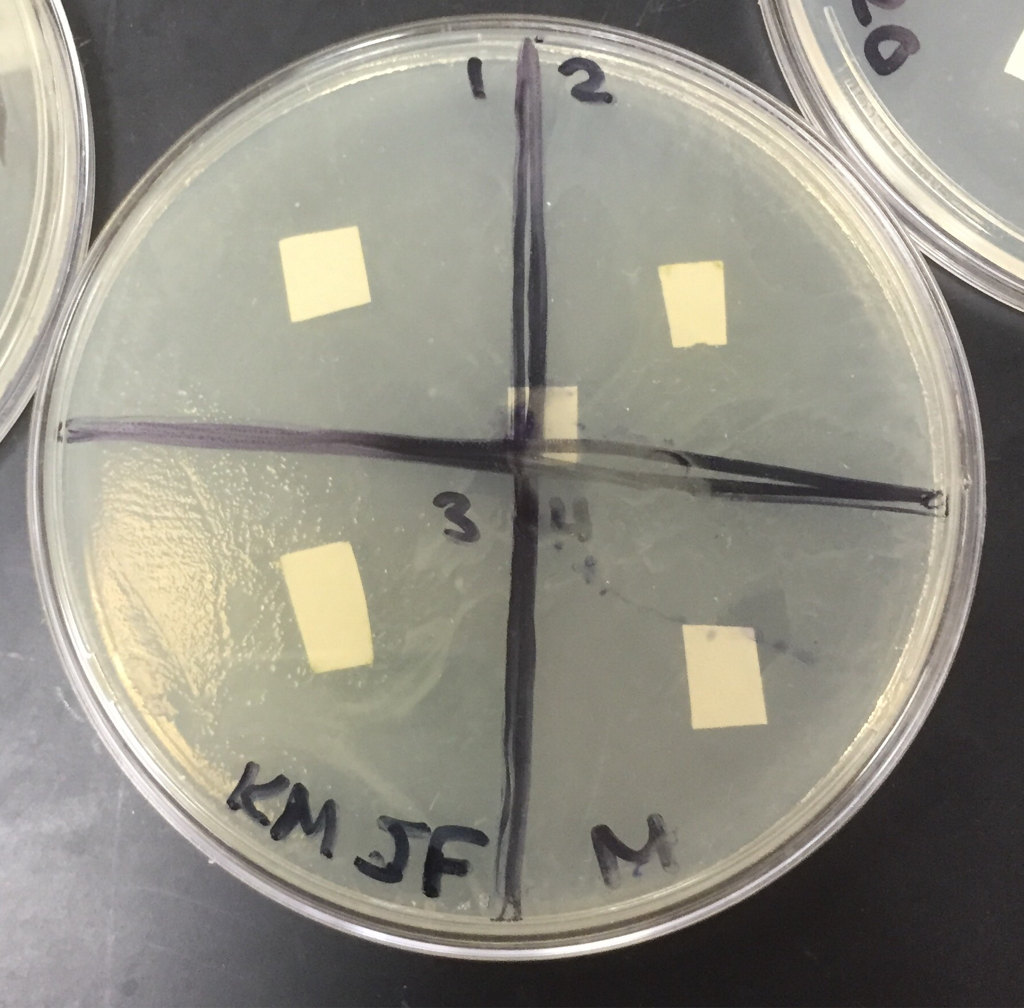
-prepare negative control disks (2-sterile water; 2 ampicillin):
- draw a cross on each plate bottom and number the quadrants 1-4
- liquids sterile LB agar in the microwave
- using sterile technique, pour approximately 20 agar into petri plate
- using sterile forceps, add the appropriate number of sterile disks to each tube of filtered extract
-prepare negative control disks (2-sterile water; 2 ampicillin):
- place the disks into the appropriate solution
Day1: No bacteria was forming as we could see, we will check again tomorrow
day 2: Bacteria is starting to form but not very noticeable we will check back tomorrow